

WORLD CUSTOMS ORGANIZATION
ORGANISATION MONDIALE DES DOUANES
Established in 1952 as the Customs Co-operation Council
Créée en 1952 sous le nom de Conseil de coopération douanière
HARMONIZED SYSTEM
COMMITTEE
NC0484E1
- O. Eng.
28th Session
-
Brussels, 20 September 2001.
POSSIBLE AMENDMENT OF THE EXPLANATORY NOTES TO CLARIFY
THE CLASSIFICATION OF VITAMIN PREPARATIONS IN THE HARMONIZED SYSTEM
(Item VIII.4 on Agenda)
I. BACKGROUND
1. The Secretariat receives numerous enquiries concerning the classification of
“vitamin preparations” every year. Similar questions are routinely received at HS seminars,
which the Secretariat organizes in different parts of the world. In order to ensure the correct
and uniform classification of such products, the Secretariat believes that an improvement of
the Explanatory Notes could be a step in the right direction.
II. SECRETARIAT COMMENTS
2. As a first move, the Secretariat is of the opinion that the classification of a specific
vitamin preparation by the HS Committee would be an appropriate first step in this process.
From there, based on the Committee’s decision and instructions, the Secretariat could
prepare draft amendments to the Explanatory Notes (in addition to a possible Classification
Opinion) to be examined at the Committee’s next session.
3. The Secretariat has selected the following product to be considered by the
Committee. This product is, in the Secretariat’s view, a typical product for this category of
vitamin preparations :
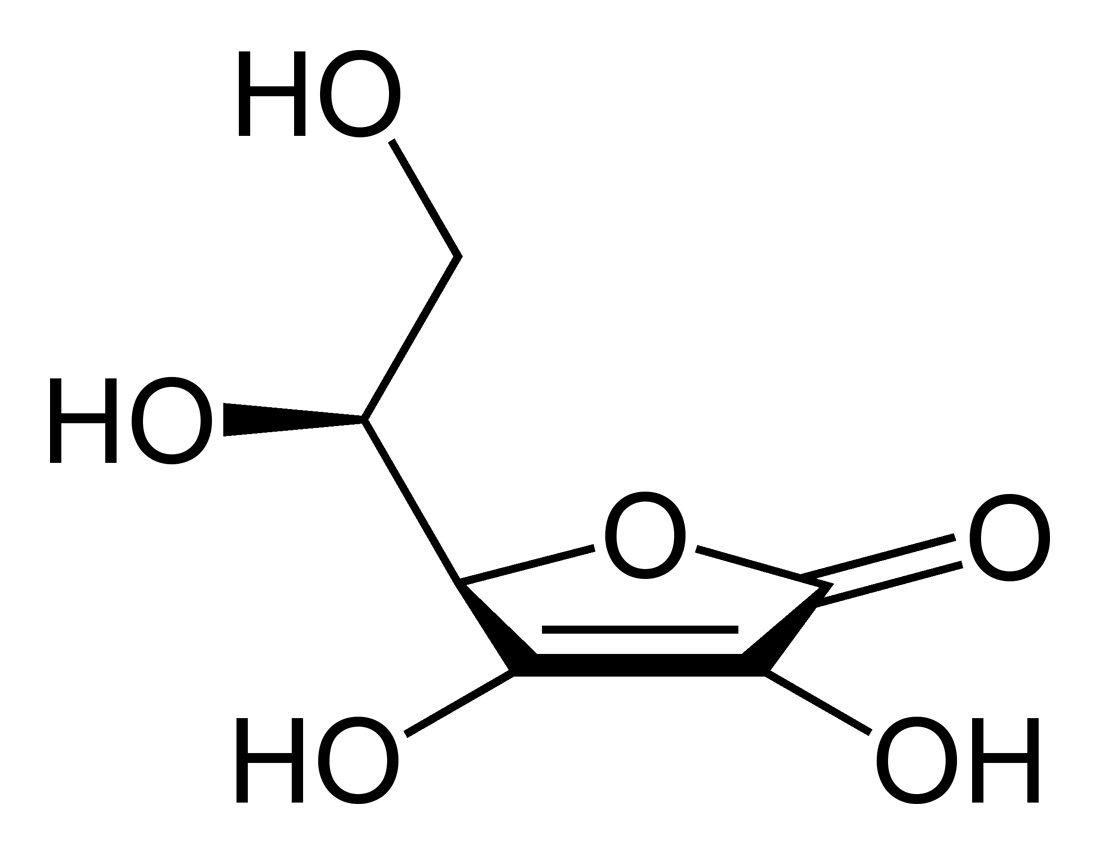
4. “*** Vitamin C 500 mg Supplement with Rose Hips” (tablets).
According to the label on the container, one tablet contains 500 mg of ascorbic acid,
corn starch, croscarmellose sodium, cellulose, rose hips, stearic acid, lemon bioflavonoid
complex, magnesium stearate and acerola. It is further printed on the label that Vitamin C is
a powerful antioxidant that may keep you healthy by neutralizing free radicals and boosting
the immune system. The tablets are put up in containers (130 tablets). The recommended
dose is one tablet daily with a meal. The following text is also printed on the label :
NC0484E1
2.
"These statements have not been evaluated by the Food and Drug
Administration. These products are not intended to diagnose, treat cure or
prevent any disease."
Classification of vitamins/vitamin preparations
5. The classification of vitamins and vitamin preparations can, in the Secretariat’s
view, be summarized as follows :
6. Vitamins/vitamin preparations can be classified in heading 21.06, 29.36 or
30.03/30.04, depending on the composition, use, presentation, etc.
7. Heading 29.36 covers vitamins and intermixtures thereof. The scope of the heading
is governed by Notes 1 (c) to (g) of Chapter 29, which stipulate that the heading is restricted
to pure vitamins or intermixtures thereof and such products (i) “dissolved in water”, (ii)
“dissolved in other solvents …”, (iii) “stabilised for the purposes of preservation or transport
by adding anti-oxidants, anti-caking agents, etc…” (see also the Explanatory Note to
heading 29.36, Item (d) on page 423), and (iv) “with an added anti-dusting agent or a
colouring matter or a odoriferous substance …”.
8. Consequently, preparations containing ingredients (other than vitamins) which are
not allowed according to the provisions of Note 1 to Chapter 29, e.g., minerals are excluded
from heading 29.36.
9. Since vitamin preparations are usually put up in measured doses and in packings
for retail sale, the question to be considered is whether they should be classified in
heading 30.04 by virtue of Note 2 to Section VI.
10. While it can be argued that vitamin preparations have prophylactic value, they are
not normally medicaments used for the prevention of a specific disease or ailment (based on
the inquiries the Secretariat has received lately). The Secretariat has examined the
classification of several vitamin/mineral preparations in the past. Such preparations basically
contain the recommended daily dietary allowances of vitamins/minerals and are therefore, in
the Secretariat’s view, food preparations which are put up and consumed, for the purpose of
maintaining general health or well-being. Vitamin preparations used in the prevention or
treatment of a specific disease or ailment would normally contain a much higher dose of an
active ingredient than the normal dietary allowance.
11. Furthermore, the HS Committee at its 21st Session (March 1998) (Doc. 42.100,
Annex F/4) agreed that the scope of headings 30.03 (and 30.04) should be limited to
products used in medicine which contain, per dose, a sufficient quantity of an active
substance with a curative or prophylactic effect against a particular ailment or disease. The
determination of active substances could be based on details made available at the time of
importation by laboratory analysis.
12. Consequently, the Secretariat would be inclined to classify most vitamin
preparations in heading 21.06, by application of Note 1 (a) to Chapter 30. This would be
supported by the Explanatory Note to heading 30.04 (page 470, second paragraph) which
excludes “food supplements containing vitamins or mineral salts which are put up for the
purpose of maintaining health or well being …” from that heading.
NC0484E1
3.
Classification of the product in question
13. The product at issue (“Vitamin C 500 mg Supplement with Rose Hips”) does not
contain minerals. However, in addition to binders, mould release agents, etc, it also contains
rose hips and acerola which are fruits known as natural sources of vitamin C for nutritional
supplements. Accordingly this product can not be classified in heading 29.36 (see Note 1 to
Chapter 29).
14. The printing on the label (“These products are not intended to diagnose, treat cure
or prevent any disease”) clearly indicates that this product does not contain, per dose, a
sufficient quantity of an active substance with a curative or prophylactic effect against a
particular ailment or disease. Consequently, this product is excluded from headings 30.03
and 30.04 (see paragraphs 11 and 12 above).
15. Accordingly, the Secretariat is of the opinion that “*** Vitamin C 500 mg Supplement
with Rose Hips” should be classified in heading 21.06, by application of Note 1 (a) to Chapter
30.
III. CONCLUSION
16. The Committee is invited to rule on the classification of a vitamin preparation
referred to as “*** Vitamin C 500 mg Supplement with Rose Hips”, on the basis of the
Secretariat’s comments set out in paragraphs 3 to 15 above.
17. The Committee is also invited to instruct the Secretariat as to what further action
(e.g., further clarification of the Explanatory Notes) should be taken in this regard (see
paragraph 2 above).
Vitamin C preparation (500 mg) in the form of tablets containing ascorbic acid, corn starch,
croscarmellose sodium, cellulose, rose hips, stearic acid, lemon bioflavonoid complex,
magnesium stearate and acerola. The tablets are put up in containers (130 tablets).
Tengo un envase de unas vitaminas con minerales con gordo lobo y extracto de semilla de uva. En la etiqueta tiene impreso “suplemento alimenticio”, la información nutricional, o sea los ingredientes en mg., la vía de administración y las instrucciones de consumo, y una leyenda que dice, “este producto no es un medicamento”, aspecto muy importante para determinar que no es para usos terapéuticos o profilácticos (p. 30.04). Existen productos que contienen vitaminas y minerales, las vitaminas son compuestos orgánicos, pero los minerales no lo son, este producto se excluye del capítulo 29. La Nota 1 del capitulo 29 establece: Salvo disposición en contrario, las partidas de este Capítulo comprenden solamente: a) los compuestos orgánicos de constitución química definida presentados aisladamente, aunque contengan impurezas; La partida 29.36 comprende c) Las mezclas de vitaminas entre sí, de provitaminas o de concentrados. en este sentido, los productos de esta partida PUEDEN ESTABILIZARSE PARA HACERLOS APTOS PARA LA CONSERVACIÓN Y EL TRANSPORTE: A CONDICIÓN de que la cantidad de sustancias añadidas o los tratamientos sufridos no sean superiores a los necesarios para la conservación o el transporte de estos productos y que esta adición o estos tratamientos NO LES MODIFIQUEN EL CARÁCTER DE PRODUCTO DE BASE Y NO LO HAGAN MÁS APTO PARA USOS DETERMINADOS QUE PARA SU UTILIZACIÓN GENERAL, así ocurre en los Productos preparados con fines terapéuticos o profilácticos, dosificados o acondicionados para la venta al por menor (partida 30.04). En mi opinión por tratarse de vitaminas y minerales, no tienen los niveles para considerar que es un producto Dosificado, es decir, que deben emplearse para fines terapéuticos o profilácticos y que debe de ser tomado bajo la dirección y supervisión de un médico; PARTIDA 21.06, PREPARACIONES ALIMENTICIAS NO EXPRESADAS NI COMPRENDIDAS EN OTRA PARTE. Esta partida comprende, entre otros: 16) Las preparaciones frecuentemente conocidas con el nombre de “COMPLEMENTOS ALIMENTICIOS” a base de extractos de plantas, concentrados de frutas u otros frutos, miel, fructosa, etc., con adición de vitaminas y, a veces, cantidades muy pequeñas de compuestos de hierro. Estas preparaciones suelen presentarse EN ENVASES INDICANDO QUE SE DESTINAN A MANTENER EL ORGANISMO EN BUEN ESTADO DE SALUD. Se excluyen de esta partida las preparaciones análogas destinadas a prevenir o tratar enfermedades o afecciones (partida 30.03 o 30.04); EL CAPÍTULO 30 PRODUCTOS FARMACÉUTICOS, en su nota 1 establece que no comprende: a) los alimentos dietéticos, alimentos enriquecidos, alimentos para diabéticos, COMPLEMENTOS ALIMENTICIOS, BEBIDAS TÓNICAS y el agua mineral, excepto las preparaciones nutritivas para administración por vía intravenosa (Sección IV); PARTIDA 30.04 COMPRENDE: MEDICAMENTOS (excepto los productos de las partidas 30.02, 30.05 o 30.06) constituidos por productos mezclados o sin mezclar, preparados para USOS TERAPEUTICOS O PROFILACTICOS, DOSIFICADOS (incluidos los administrados por vía transdérmica) o acondicionados para la venta al por menor, en este sentido, Se consideran como tales los productos que por su acondicionamiento y principalmente por la PRESENCIA EN CUALQUIER FORMA DE INDICACIONES APROPIADAS (naturaleza de las afecciones contra las que deben emplearse, modo de usarlos, posología, etc.) son identificables como destinados a la venta directa a los usuarios sin otro acondicionamiento (particulares, hospitales, etc.), para utilizarlos con los fines anteriormente indicados. LAS DISPOSICIONES ENUNCIADAS EN EL TEXTO DE LA PARTIDA NO SE APLICAN a los alimentos ni a las bebidas (tales como: alimentos dietéticos, alimentos enriquecidos, alimentos para diabéticos, bebidas tónicas y aguas minerales naturales o artificiales), que siguen su propio régimen. Tal es, esencialmente, el caso de LAS PREPARACIONES ALIMENTICIAS QUE SÓLO CONTIENEN SUSTANCIAS NUTRITIVAS. Los elementos nutritivos más importantes contenidos en los alimentos son las proteínas, los carbohidratos y las grasas, LAS VITAMINAS Y LAS SALES MINERALES también desempeñan un papel en la alimentación. Lo mismo ocurre con los alimentos y bebidas con sustancias medicinales agregadas, desde el momento en que estas sustancias no tengan otro propósito que EL DE CREAR UN MEJOR EQUILIBRIO DIETÉTICO, AUMENTAR EL VALOR ENERGÉTICO O NUTRITIVO DEL PRODUCTO O MODIFICAR EL SABOR, SIEMPRE QUE NO PRIVEN AL PRODUCTO DEL CARÁCTER DE PREPARACIÓN ALIMENTICIA. Por otra parte, los productos consistentes en una mezcla de plantas o partes de plantas solas o mezcladas con otras sustancias, empleadas para hacer infusiones o “tisanas” (por ejemplo, aquéllas que tienen propiedades laxantes, purgantes, DIURÉTICAS o carminativas), INCLUIDOS LOS PRODUCTOS QUE OFRECEN ALIVIO A DOLENCIAS O CONTRIBUYEN A LA SALUD Y BIENESTAR GENERAL, también se excluyen de esta partida (partida 21.06). Además, esta partida (30.04) no comprende LOS COMPLEMENTOS ALIMENTICIOS QUE CONTENGAN VITAMINAS O SALES MINERALES, que se destinen a conservar el organismo en buen estado de salud, PERO QUE NO TENGAN INDICACIONES RELATIVAS A LA PREVENCIÓN O AL TRATAMIENTO DE UNA ENFERMEDAD. Estos productos, que se presentan comúnmente en forma de líquidos, pero que pueden presentarse también en polvo o en comprimidos, se clasifican generalmente en la partida 21.06 o en el Capítulo 22. Por el contrario, quedan clasificadas aquí (p. 30.04) las preparaciones en las que las SUSTANCIAS ALIMENTICIAS O LAS BEBIDAS SE UTILIZAN COMO SIMPLE SOPORTE, excipiente o edulcorante DE LAS SUSTANCIAS MEDICINALES, principalmente para facilitar su ingestión. Hay que verificar la información de las etiquetas de los productos que deben de contener la información que menciono al inicio. Esta clase de productos son normalmente comercializados como suplementos alimenticios o nutricionales y no tienen indicaciones que son para usos profilácticos o terapéuticos.
AGRADECIMIENTO AL V.A. OSCAR ALFARO, POR SU APORTE.

..bmp)




..jpg)
..jpg)







.jpg)
.gif)
.jpg)









..jpg)

+Stainless+steel+elbow.bmp)



..jpg)

..jpg)

..jpg)



















..jpg)
..jpg)


..jpg)







Tablestacas..gif)

.gif)
.bmp)











..jpg)



.jpg)



..jpg)



.jpg)




..jpg)



.gif)




..jpg)


















+de+madera+para+vias,sin+impregnar.jpg)








..jpg)



.gif)


..jpg)
+de+oruga.jpg)


















+BEL-ART.jpg)











.jpg)
..jpg)
.gif)
..bmp)





.bmp)


No hay comentarios:
Publicar un comentario